Introduction
Materials and Methods
Preparation of plant material and generation of crosses
Crossing procedure
Seedlings preparation
Field layout and situation
Studied characters
Statistical analysis
Results
Mean performance of estimated traits
Analysis of variance
ANOVA for L × T analysis
Gene action and contribution of line, tester and line x tester interaction
Correlation analysis
Combining ability
Discussion
Introduction
Lily, one of the economically important flower bulbs, belongs to genus Lilium which comprises of 110 species (McRae, 1998), 7 sections and 10,000 documented cultivars (De Jong, 1974; Mathews, 2007; Bakhshaie et al., 2016). Many commercial cultivars have been produced by interspecific hybridization (van Tuyl and Arens, 2011) and propagated by vegetative methods (McRae, 1998).
The Lilium × formolongi hort; an interspecific hybrid of L. formosanum and L. longiflorum, is a popular commercial cut flower in Korea, Japan and China (Wadanabe, 1988a, 1988b; Kunishige, 1993; Watanabe, 1993; Xuan et al., 2004; Ho et al., 2006). L. × formolongi is mainly seed propagated, seeds sown during december to february flowered in august (Goo et al., 1995, 2003; Xuan et al., 2005).
One of the common methods of L. × formolongi seed production is bulk population seed system (Watanabe, 1988b). Though there are various modified method about bulk population seed production, it is necessary to fix the transgressive segregates to reach near the level of homozygosity (Xuan and Kim, 2014). On the other hand while using the F1 seeds in L. × formolongi, the stability of novel cut flower traits is a major concern for commercial scale production (Ho et al., 2006). There are several disadvantages associated with seed propagation method viz. the time required for the plants to reach a suitable sized bulb for cut flower production, undesirable variability and seasonal supply (Bakhshaie et al., 2016). It is obvious that populations derived from the seed propagation are not uniform in plant height, flowering time and flower shape (Roh, 2002), ultimately increases the cut flower production cost due to the unwanted labor cost. For example, populations derived from seed propagation demonstrated spreading flowering time more than one month thereby increasing labor cost. In this regard, breeding should focus on the uniformity of seedling populations. So alternative production methods need to be developed to shorten the total production time focusing on the induction of early flowering with keeping potentiality of the bulbs to produce the maximum number of flower buds constant. Tissue culture and scaling are a preferable method to produce the clonal line to maintain the heterozygous genotypes for the seed production of homogenous populations as clonal lines are genetically as well as phenotypically identical.
In this experiment, we have tried to make clonal parental line and testers through scaling and mated them in the design of Line × Testers mating system to understand the combining ability and gene action for the inheritance of commercial quantitative traits.
Materials and Methods
Preparation of plant material and generation of crosses
On the basis of morphological performance, 3 promising single cross F1s of L. × formolongi and 6 CVs/breeding lines (2 CVs and 1 breeding line were represented from L. × formolongi and 1 CV and 2 breeding lines were represented from L. Longiflorum) (As shown in Table 1) were selected and scaling has been observed taking 20 lines from each genotype. Scaling has been conducted following the standard scaling techniques. One clonal line has been selected from each genotype. Selected clonal lines of three single cross F1s were used as lines and selected clonal lines of 6 CVs/breeding lines were used as testers. All 9 clonal lines have been crossed in Line × Testers mating design to produce 18 L × T hybrids. In this way genotypically resulting progenies were three-way cross F1s (3WCF1s). Altogether 27 genotypes (including parents) had been prepared for the study. The testers (donors) parents were purposefully selected to improve some important cut flower traits which are lagging in female (lines) parents. Sib crosses among the selected clonal lines have been made to maintain seed lots of the parental clonal lines.
Table 1. Name and pedigree history of L. × formolongi parental genotypes
Crossing procedure
Mother bulbs of testers (remaining after peeling out the matured scale) has been planted in planting boxes for the purpose of collection of pollen. One day before to anthesis in the evening anthers has been collected in the petri dish and dried overnight under light of mercury tube and kept at 4°C inside the refrigerator for long-term future use. Emasculation of the female parent was done just 1 day before anthesis in the evening and plants were tagged after capping the stigma separately. Next day in the morning, dried pollen of male parent had been put on (attached) the stigma of the selected female parent using forceps for hybridization. Name tag over the crossed plant has been provided denoting in details, the date of crossing, name of female parent, name of the male parent and number of ovary crossed etc.
Seedlings preparation
Prepared seeds of all 27 genotypes have been treated with the recommended fungicide for 24 hrs. Then after cold treatment has been provided to the seeds up to 2 weeks keeping inside the refrigerator at 4°C. In the first week of February 300 seeds of each genotype have been sown over the trays inside plastic house. 2/3rd a portion of trays (size 60-40-6) has been filled with recommended soil mixed with the recommended dose of fertilizers and insecticides and seeds were uniformly shown over the trays 300 seeds/tray; mixing properly with fine sand or fine particles obtained after sieving of vermiculite. Then seeds were covered properly using vermiculite and provided water properly using tap nozzle and finally covered all the trays using a thin white plastic sheet to maintain the moisture. The small plastic tunnel has been made using the iron rod of 2 m length and inside tunnel bulb light has been fixed across the vertical bar of semi-circle iron rod. Inside plastic house during daytime temperature has been maintained 25 ± 3°C and night temperature has been maintained 15 ± 3°C using bulbs light and heater. After 2 weeks when 2/3rd (70%) seeds were germinated; plastic cover has been removed. Timely irrigation (3 times/week) and insecticide, fungicide and fertilizers are applied as per need.
Field layout and situation
All plant material (27 genotypes) including 9 parents and 18 L × T hybrids were grown in Randomized Complete Block Design (RCBD) with three replications maintaining 240 populations of each genotype for evaluation of studied traits in summer season in 2014 at the farm of KNU, Horticulture department (Floricultural breeding laboratory), Chuncheon, Kangwon-do, South Korea. The genotypes were grown in multiple bed plots; Each bed was 30 m long, 1 m in width; seedlings were transplanted maintaining row to row and plant to plant equal distance of 12.5 cm. Standard seedlings growing cultural practices as adopted by local lily grower farmers has been adopted to obtain the healthy flower plants to achieve the good results.
Studied characters
Morphological observation for 10 quantitative traits has been taken; sampling 4 plants from each replication. Plant height was measured in centimeter (cm), stem diameter in millimeter (mm.), number of leaves per plant in numbers (nos.), leaf length in centimeter (cm), leaf width in centimeter (cm), days to flowering in number of days (days), number of flowers per plant in numbers (nos.), flower diameter in in millimeter (mm.), bud length in centimeter (cm) and attitude of the floral axis/flower direction in degree (°).
Statistical analysis
The data recorded on all genotypes; 3 lines, 6 testers and 18 L × T hybrids were analyzed using MS-Excel-2013 and TNAUSTAT statistical packages. General ANOVA for parents and hybrids, ANOVA for line × tester analysis, estimation of components of genetic variances, estimation of combining ability effects (general combining ability effects and specific combining ability effects), proportional contribution of lines, testers and their interactions to total variance and correlation coefficient (both genotypic and phenotypic) were estimated using the TNAUSTAT statistical packages (https://sites.google.com/site/tnaustat/plant-breeding-heterosis).
Manual estimation of these parameters has been outlined by Nadarajan et al. (2016) in Quantitative genetics and Biometrical techniques in Plant Breeding. Ranking of the best specific combiner has been done on the basis of overall GCA status of their parents using the method as outlined by (Arunachalam and Bandyopadyya, 1979).
Results
Mean performance of estimated traits
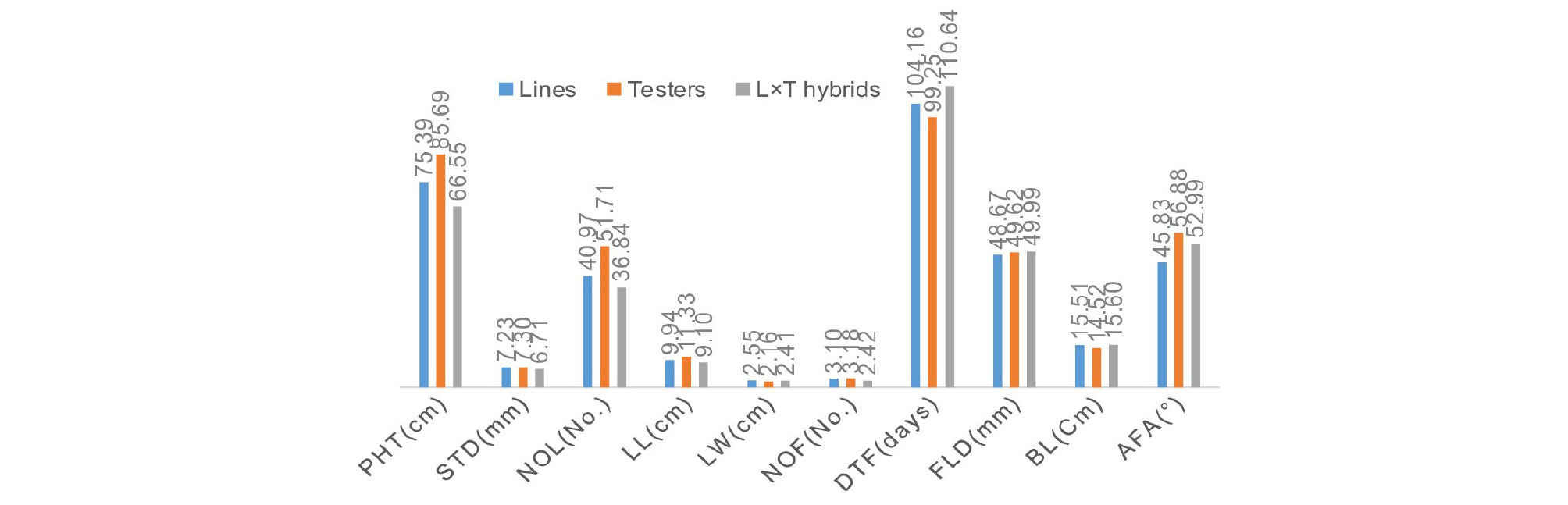
Mean performance of lines (SCF1s), testers (CVs/breeding lines) and their L × T hybrids demonstrated genetic variability among the studied quantitative traits. Mean performance of testers were found to be highest for 7 traits except for leaf width, flower diameter and bud length. Likewise mean performance of L × T hybrids were superior for most floral traits like flower diameter, bud length and attitude of the floral axis. Mean performance of lines were found to be intermediate for almost studied quantitative traits (Fig. 1).
Analysis of variance
The mean sum squares of all studied traits are given in ANOVA Table 2. There were observed significant difference among the genotypes for all the studied traits. Parent & Cross were significantly different for all 10 quantitative traits. Cross vs. Parent were significantly different for almost traits (except leaf width) indicating the presence of heterosis for these traits.
Table 2. ANOVA for mean sum squares for 1o quantitative traits in L. × formolongi
ANOVA for L × T analysis
The mean sum squares for all the studied traits for lines, testers and L × T interaction are given in Table 3. The lines and Testers are significantly different for all studied traits. Likewise, L × T interaction indicates significantly different for almost traits except for attitude for floral axis.
Table 3. ANOVA for combining ability of quantitative traits of parents and hybrids in L. × formolongi
Gene action and contribution of line, tester and line x tester interaction
Some of the traits viz. plant height, leaf length, days to flowering and attitude of floral axis showed prevailing of additive type of gene action, while rest of the traits i.e. stem diameter, number of leaves, leaf width, number of flower, flower diameter and bud length showed predominantly dominance gene action (Table 4).
Table 4. Estimated variance of GCA and SCA for the studied character
The contribution of line, testers and L × T interaction for variation of quantitative traits of L × T hybrids are shown in Table 5. The contribution of lines was found 92.35% highest for plant height and 0.91%lowest for diameter of flower. Then contribution of testers was found 80.67% highest for leaf width and 2.29% lowest for plant height. Likewise, the contribution of L × T interaction ranged from 4.51% to 42.02% lowest to highest range for the trait of attitude of floral axis and number of flower respectively.
Table 5. Contribution of Lines, Testers and L × T hybrids for 10 quantitative traits in L. × formolongi
Correlation analysis
Genotypic correlation coefficient and phenotypic correlation coefficient among the 10 quantitative traits of all 27 genotypes (3 lines, 6 testers and 18 L × T hybrids) has been estimated employing the replication wise mean value.
Phenotypic and genotypic correlation coefficient (Table 6) indicated most of the quantitative traits are correlated with other quantitative traits. Both genotypic and phenotypic correlation coefficient indicated that plant height highly correlated with stem diameter, number of leaves, leaf length, and number of flower at 1% level of significance and correlated with attitude of floral axis at 5% level of significance.
Table 6. Genotypic correlation (above diagonal) and phenotypic correlation (below diagonal) among 10 quantitative traits in L. × formolongi
Combining ability
Two types of combining ability; 1.General combining ability (GCA) and 2.Specific combining ability (SCA) usually have been discussed in quantitative genetics.
General combining ability effects
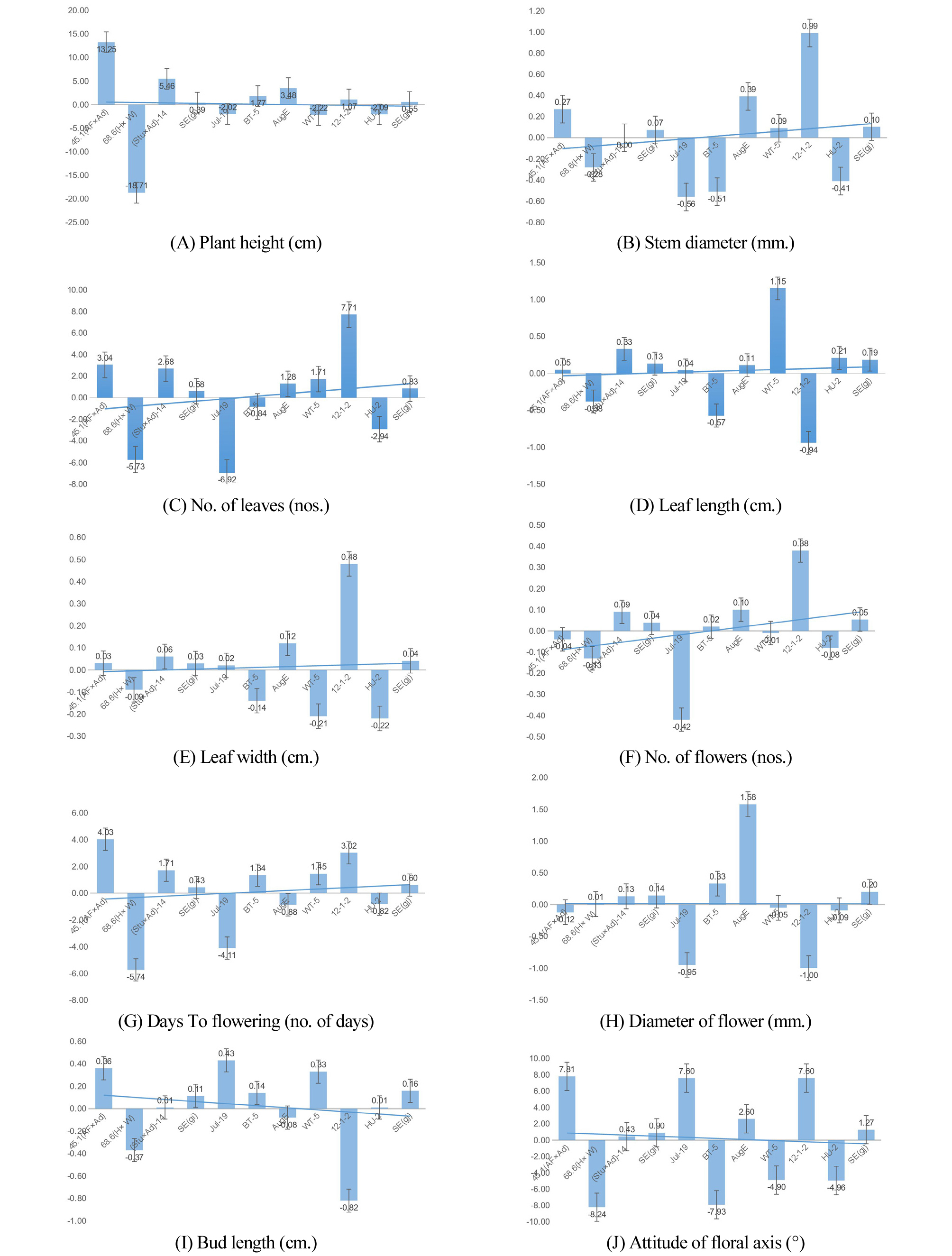
GCA effects are in general outcomes of additive type of gene action. The estimated value of GCA effects of lines and testers are given in (Fig. 2(A)-(J)). Among the three lines, on the basis of GCA effects two lines; lines 1, 45-3 (AF × Ad) and line 3, (Stu × Ad) -14 demonstrated superior performance with significant GCA effects for 5 traits viz.plant height, stem diameter, number of leaves, bud length, attitude of the floral axis of lines 1 and plant height, number of leaves, leaf length, leaf width and number of flower of line 3.
Among the testers 3 parental lines demonstrated superior performance with significant GCA effects ;tester 3 possessed significant GCA effects for 5 traits (PHT, STD, LW, FLD and AFA), tester 5 possessed significant GCA effect for 5 traits (STD, NOL, LW, NOF and AFA)and tester 1 possessed significant GCA effects for 3 traits (DTF, BL and AFA).
Specific combining ability (SCA) effects
SCA is defined as the deviation in the performance of hybrids from the expected performance based on the average performance of lines involved in the hybrid combination. SCA effects included both dominance and epistatic gene effects. It can be used as a basic indicator of superiority of particular cross combination for particular traits. Since none of the hybrids possessed significant SCA effect for all studied quantitative traits; best specific combiner along with GCA status of their parents, mean performance and combination status for all the traits have been presented in Table 7.
Table 7. Ranking of superior specific combiner for particular quantitative traits
L × T hybrid (3 × 1) demonstrated significant SCA effects for the 3 traits viz. plant height, stem diameter and bud length. Likewise, L × T hybrid (2 × 6) demonstrated significant SCA effects for the 3 traits viz. plant height, leaf width and number of flower; demonstrating their superiority for that traits.
Discussion
Lilium × formolongi is a self-pollinating bulbous crop usually used for cut flowers purpose. The line × tester analysis is used in both self as well as cross-pollinated crops (Kempthorne, 1957).
The mean performance of lines, testers and L × T hybrids demonstrated significant variability for studied quantitative traits (Fig. 1). The foundation of variation is due to genotype (genetic constitution) of lines (single cross F1), testers (commercial cultivar and breeding lines) and L × T hybrids (3WCF1). The mean performance of quantitative traits of L × T hybrids represented the contribution of lines, testers and Line × Testers interaction as a whole (Table 5). The performance of SCF1 (lines) was found superior for almost of traits viz. plant height, stem diameter, number of leaves, leaf length, leaf width, number of flowers, days to flowering as compared with 3WCF1 (L × T hybrids) while the performance of 3WCF1s was found superior for most of the floral traits viz.flower diameter, bud length and attitude of the floral axis as compared with SCF1 (lines).
On the other hand the mean performance of testers demonstrated superior performance over SCF1s and 3WCF1s for the traits of plant height, stem diameter, number of leaves, leaf length, number of flower and days to flowering etc. thereby providing rationality for selecting testers to improve these traits.
The mean performance also indicated that line1, 45-1(AF × Ad) was superior for plant height, stem diameter, leaf length, number of flowers, bud length and attitude of the floral axis. Likewise, parent-2, (Stu × Ad)-14 found to be superior for the number of leaves, days to flowering and flower diameter etc. Plant height, the number of flower and attitude of the floral axis are the quantitative traits deserving prime importance for the purpose of cut flower. Both of the female parents we can use for homogenous seed production in plastic house.
Similarly in case of testers, out of six testers, tester-1, Julius-19 has demonstrated significant performance for leaf length, leaf width, days to flowering, bud length and attitude of the floral axis. It could be the better donor for selected female parents viz. 45-1 (AF × Ad) and (Stu × Ad)-14 for seed production inside the plastic house. Among 18 hybrids evaluated, none of the crosses were superior for all the studied traits. Among the L × T hybrids (3 × 1) demonstrated significant SCA effects for the 3 traits viz. Plant height, stem diameter and bud length. Likewise, L × T hybrid (2 × 6) demonstrated significant SCA effects for the 3 traits viz. Plant height, leaf width and the number of flowers; demonstrating their superiority for that traits. These genotypes can exploit the further breeding program or some selected lines directly can use as the cultivar. Montazeri et al. (2014), has found variability among the agronomic traits of lines, tester, and hybrids in Rice. Furthermore, Kumari et al. (2015) has also found significant variation among the character association of agronomic and biochemical traits of lines, testers, and hybrids in Pea.
The next and most important insight into the combining ability analysis is related to GCA and SCA and its variances which represents the relative measure of additive and non-additive gene actions involved in the inheritance of quantitative traits respectively. Plant height, leaf length, days to flowering and attitude of the floral axis (Song et al., 2004) showed prevailing of the additive type of gene action, opening the possibility for backcross breeding with a potential donor for those traits for the improvement of those traits. But with contrast to the findings of Xuan et al. (2005), rest of the traits i.e. Stem diameter, number of leaves, leaf width, number of flowers, flower diameter and bud length showed predominantly dominance gene action. These quantitative traits emphasized the importance of heterosis breeding (Kumari et al., 2015). The combining ability analysis demonstrated significant differences due to interactions of line x tester for all traits except leaf length, thereby suggesting the importance of both additive and non-additive variance for these traits (Bagheri and Babaeian, 2010). In this context, the study of both additive and non-additive gene actions required for improving related-quantitative traits. Moreover, the contribution of line × testers interaction has shown large margin for the traits Stem diameter (20.94%), the number of flowers (42.02%), Flower diameter (32.90%) and bud length (30.33%) demonstrating the importance of non-additive gene action for these traits.
Correlation among the studied quantitative traits is another one of the most important insights into the process of selection. Combining ability with gene action for the inheritance of particular quantitative traits in combined with correlation among the traits helps breeder for the process of judicious progeny selection. In this experiment correlation analysis among the studied quantitative traits demonstrated that plant height correlated with stem diameter (Song et al., 2004), number of leaves and number of flower (Xuan et al., 2005). In addition to the finding of Xuan et al. (2005), plant height was also correlated with leaf length, and attitude of the floral axis.
The SCA effects represent both dominance and epistatic effects of a particular cross combination which we can consider as an indicator to determine the usefulness of that particular cross combination for exploitation through heterosis breeding and hybridization followed by recombination. The result of SCA effects indicated that none of the cross combination has possessed significant SCA effects for all quantitative traits studied. The high specific combiner involved (high × high), (high-low) and (low × low) general combiner as parents provided the clear evidence that high performance of these crosses may be attributed to additive × additive, additive × dominance or dominance × dominance (low × low) epistatic interactions. Thus high GCA effects of the parents, therefore, do not seem to be a reliable criterion for the prediction of high SCA effects. Similar results have been reported by Kumari et al. (2015) in Pea. Narasimhamurthy and Gowda (2013), Sharma et al. (2013) had also reported that the good specific combiners for different characters involved parents with (high × high), (high-low), (low × high) and (low × low) general combination. In majority of the cases, the crosses exhibiting high SCA effects were found to have both or one of the parents as good general combiners for the characters studied revealing non-additive gene action in the genetic control. Sharma et al. (2013), in their review about the combining ability and gene action in garden pea, has also concluded in line with this insight. It is visibly evident that due to the lack of complementation of the parental genes, those parents having high GCA effects produced low SCA effects for some traits. Likewise, for some traits, parents having low GCA effects produced hybrids with high SCA effects due to the complementary gene action (Kumari et al., 2015).