Introduction
Materials and Methods
Chemicals and reagents
Sprouting method and extraction
Total phenolic content
Total flavonoid content
Tannin content
DPPH radical scavenging assay
ABTS radical scavenging assay
Results
Extraction yield and total phenolic content
Flavonoid content
Tannin content
DPPH free radical scavenging activity
ABTS•+ scavenging activity
Discussion
Introduction
Vegetables play a key role in the diet of humans because of their support in the normal functioning of different body systems. It is well-known that the consumption of vegetables could reduce the risk of various chronic diseases in humans (Bjorkman et al., 2011; Vale et al., 2014). Vegetables contain several health-promoting nutrients and bioactive compounds, including polyphenolics, vitamins, carotenoids, minerals, and fiber. In particular, phenolic compounds are recognized as having valuable health benefits, because of their remarkable antioxidant properties (Moreno et al., 2006; Wei et al., 2011). Dietary phenolics can reduce the risk of various chronic diseases, such as cardiovascular disease, inflammation, cancers, and diabetes, by suppressing the production of free radicals (Kowalska et al., 2014).
In recent times, the role of Brassicaceae sprouts and vegetables in the diet of humans has gained attention because of their high nutritional value and rich bioactive metabolite contents (Vale et al., 2014). Brassicaceae vegetables have various pharmacological properties, such as antioxidant, gastroprotective, anti-inflammatory, and anti-obesity activities. These vegetables can be used in different forms, including raw, cooked, fried, baked, and fermented (Samec et al., 2017). In addition, the sprouts have received considerable attention over the past decades because of their low-fat level, which contributes to the improvement of human health (Li et al., 2018). These plants are grown worldwide their good environmental adaptation. Among the various species, Brassica oleracea is the most important member of Brassicaceae; it has great global economic importance. The consumption of Brassicaceae vegetables also significantly regulates metabolic activity in the body (Cartea et al., 2011). Flavonoids (flavonols and anthocyanins) and hydroxycinnamic acids (p-coumaric, sinapic, and ferulic acids) are the most widespread group of polyphenolic compounds in Brassicaceae vegetables (Olsen et al., 2009; Vale et al., 2015).
Edible sprouts possess higher nutritional profiles than those of vegetable products, according to several studies (Martinez-Villaluenga et al., 2008; Yuan et al., 2010). Some Brassicaceae members, such as broccoli, cabbage, and radish, are rich sources of various antioxidant compounds. These sprouts are particularly recommended as the best sources of polyphenols, glucosinolates, fiber, carotenoids, vitamins, and minerals (Nicola et al., 2013). Some health-promoting bioactive components, such as glucosinolates, are found in almost 10 times higher quantities in sprouts than in vegetables (Wei et al., 2011; Yuan et al., 2010). Previous studies found that the breakdown products of glucosinolates (isothiocyanates) have the potential to reduce the risk of different types of cancers (Nicola et al., 2013; Vale et al., 2015). Further, the consumption of sprouts stimulates the immune system activation, alleviates vitamin and mineral deficiencies, and prevents several serious illnesses (Wezyk and Kazepilko, 2012). Sprouts are an important alternative to increasing the consumption of seeds in human nutrition. However, there have been no comparative studies in relation to the phytochemical analysis and antioxidant activity of Brassicaceae sprouts and their commercial vegetables. Hence, this study aimed to evaluate the total phenolic content and antioxidant activity of selected Brassicaceae sprouts (red radish, red kohlrabi, pink and red cabbage, tatsoi, and broccoli) and their commercial vegetables.
Materials and Methods
Chemicals and reagents
1,1-diphenyl-2-picryl hydrazyl (DPPH), ascorbic acid, 2,2'‐azinobis (3‐ethylbenzothiozoline‐6‐sulfonic acid) diammonium salt (ABTS), potassium persulfate, Folin–Ciocalteu phenol reagent, sodium nitrite, catechin, aluminum chloride, sodium hydroxide, sodium carbonate, polyvinyl polypyrrolidone (PVPP), and gallic acid were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Sprouting method and extraction
Seeds of Raphanus sativus L. (red radish), Brassica oleracea var. gongylodes L. (red kohlrabi), Brassica oleracea var. capitata (pink cabbage), Brassica oleracea var. capitata L. f. rubra (red cabbage), Brassica campestris var. narinosa (tatsoi), and Brassica oleracea var. italic (broccoli) from Aram Seed Store (Seoul, Republic of Korea), and mature fresh vegetables were purchased from E-mart, Chuncheon, Republic of Korea. To obtain sprouts, seeds were grown in a hydroponic growth chamber for three weeks (Jung Hoon E & C, JH6130, Republic of Korea) at 24°C with a blue and red LED light source (24 V) (light/dark cycle of 16/8 h). The wavelength of blue light is 430–500 nm and that of red light is 600–660 nm. The flow rate of water was 30 s/90 min. After 21 days, the sprouts were cut into small pieces and freeze-dried. The ethanol (95%) extract of sprouts was obtained by the maceration technique (Tachakittirungrod et al., 2007). After three days of incubation time at room temperature, the extract was filtered using Whatman No. 1 paper and concentrated using a rotary vacuum evaporator (EYELA NE-1001, Tokyo, Japan). Subsequently, the concentrated extract was lyophilized using a freeze dryer (FD5505 SPT, Ilshin, Seoul, Republic of Korea). For the commercial vegetables, the fresh vegetables into small pieces and directly used for the extraction. The same method as that described above for sprouts was used to obtain the ethanol extract of vegetables. The extract yield obtained from Brassicaceae sprout and vegetable samples was expressed as percent of the plant sample dry matter.
Total phenolic content
The total phenolic content of extracts was determined by the Folin–Ciocalteu method modified to 96-well plate method, as described by Mokrani et al. (2016). Gallic acid was used as a standard to generate the calibration curve. Dried extracts were dissolved in ethanol (1 mg /mL). In the 96 well plate, 20 µL of extract or the standard or ethanol (blank) was added. Subsequently, 100 µL of Folin-Ciocalteu reagent (Folin-Ciocalteu reagent 1: distilled water 9) was added. After 3 minutes, 80 µL of sodium carbonate (75 g/L) solution was added. Then, the mixture was incubated in the dark at room temperature for 60 min. The absorbance was measured at 765 nm using a microplate reader (SpectraMax 190, Molecular Devices, USA). The analysis was performed in triplicate and the results were expressed as mg gallic acid equivalent per g of extract (mg GAE/g).
Total flavonoid content
The total flavonoid content of the extracts was determined according to the method described by Herald et al. (2012). Catechin was used as a standard to generate the calibration curve. A 100 µL of distilled water was added to each of the 96 wells, followed by 10 µL of sodium nitrite (50 g/L), and 25 µL of the sample or the standard solution. After 5 min, 15 µL of aluminum chloride (100 g/L) was added to the plate. After 6 min of incubation time, 50 µL of sodium hydroxide (1 M/L) and 50 µL of distilled water were added. The shaken well and the absorbance was measured at 510 nm using a microplate reader (SpectraMax 190, Molecular Devices, USA). The analysis was performed in triplicate and the results were expressed as mg catechin equivalent per g of extract.
Tannin content
The tannin content of extracts was estimated after treatment with polyvinyl polypyrolidone (PVPP) (Makkar et al., 2003). Total phenolics comprise both tannin and non-tannin phenolics. One hundred mg of PVPP was added to 2 mL Eppendorf tubes, followed by 500 µL of distilled water and 500 µL of the extract were added and vortexed the mixture well. Then, the Eppendorf tubes were incubated at 4°C for 4 h. After incubation, the sample was centrifuged at 3,000 rpm for 10 min at 4°C and the supernatant was collected. The non-tannin phenolics was determined using the same method as that described in the total phenolics section (Mokrani et al., 2016). The tannin content was calculated by subtracting the total non-tannin phenolics (free phenolics) from the total phenolics. The analysis was performed in triplicate and the results were expressed as mg GAE/g.
DPPH radical scavenging assay
The DPPH radical scavenging assay was performed according to the method described by Fu et al. (2014) with minor modification. Briefly, 100 µL of the sample or the standard (ascorbic acid) solution was mixed with 100 µL of DPPH solution (0.2 mM, in methanol). The reaction mixture was shaken and incubated for 30 min at 25°C. Then, the absorbance was measured at 517 nm using a microplate reader (SpectraMax 190, Molecular Devices, USA). The analysis was performed in triplicate and the IC50 values of the extract i.e., the concentration of extract necessary to reduce the initial concentration of DPPH by 50% was calculated.
ABTS radical scavenging assay
The ABTS radical scavenging activity was determined using the method of Fu et al. (2014) with minor modifications. Briefly, the ABTS•+ solution was produced by reacting 7 mM ABTS with 2.45 mM potassium persulfate in the dark for 12–16 h at room temperature. Prior to the assay, 1 mL of ABTS stock solution was diluted with 89 ml of absolute ethanol (1:89) and equilibrated to obtain an absorbance of 0.700 ± 0.02 at 734 nm. The absorbance was measured using a microplate reader (SpectraMax 190, Molecular Devices, USA). An aliquot of 50 µL of sample was mixed with 150 µL of ABTS solution. After the incubation period (30 min at room temperature), the absorbance was measured at 734 nm. The analysis was performed in triplicate and the IC50 values were calculated.
Results
Extraction yield and total phenolic content
The yield and total phenolic content of ethanol extracts from different Brassicaceae sprouts and commercial vegetables are shown in Table 1. The ethanol extracts from all of the sprouts gave a higher yield than did those from the vegetables. In these, we obtained the maximum extraction yield in red radish sprouts (25.03%). Among the six sprouts, broccoli showed the highest total phenolic content (25.750 mg GAE/g extract), followed by red cabbage, pink cabbage, red kohlrabi, and red radish sprouts. Sprouts from tatsoi presented the lowest total phenolic content (6.522 mg GAE/g extract). For commercial vegetables, tatsoi (31.511 mg GAE/g extract) and pink cabbage (2.141 mg GAE/g extract) presented the highest and lowest total phenolic contents, respectively. Compared to those of vegetables, all sprouts, with the exception of tatsoi, exhibited significantly (p < 0.05) higher total phenolic contents.
Table 1.
The yield and total phenolic, flavonoid, and tannin contents of Brassicaceae sprouts and their commercial vegetables
Flavonoid content
Similar to the total phenolic content, the total flavonoid content of sprouts was higher than those of vegetables, with the exception of tatsoi (Table 1). We found the highest and lowest total flavonoid content in red cabbage sprouts (14.261 mg catechin/g extract) and tatsoi sprouts (2.904 mg catechin/g extract), respectively. Among the six vegetables, tatsoi showed the highest flavonoid content (15. 845 mg catechin/g extract), followed by broccoli and red cabbage. In addition, the extracts from the vegetable and sprouts of broccoli contained similar flavonoid contents (11.380 mg catechin/g extract). Red radish, red kohlrabi, and pink cabbage vegetables contained relatively low flavonoid content.
Tannin content
The tannin content of Brassicaceae sprouts and their commercial vegetables is presented in Table 1. All sprouts had lower tannin content (0.431–2.729 mg GAE/g extract) with the exception of those of broccoli (8.068 mg GAE/g extract). Similar to the total phenolic and flavonoid contents, we observed the highest tannin content in the tatsoi vegetable (11.649 mg GAE/g extract). Further, tatsoi and red cabbage (11.626 mg GAE/g extract) vegetables showed notably similar tannin content. We found the lowest tannin content in the pink cabbage vegetable (0.285 mg GAE/g extract), followed by the red radish vegetable (0.979 mg GAE/g extract).
DPPH free radical scavenging activity
The results of DPPH free radical scavenging activity of Brassicaceae sprouts and their vegetables are depicted in Fig. 1. Except for tatsoi sprouts, the ethanol extracts of all sprouts showed higher DPPH radical scavenging activity than those of their respective vegetables. Among different sprouts, red radish sprouts and tatsoi sprouts exhibited the highest and lowest DPPH scavenging activity, respectively. The DPPH free radical scavenging effect of sprouts was in the following order: red radish (IC50 of 358 µg/mL) > red cabbage (391 µg/mL) > pink cabbage (428 µg/mL) > red kohlrabi (452 µg/mL) > broccoli (520 µg/mL) > tatsoi (1,053 µg/mL). For the vegetables, the ethanol extract from red cabbage registered the highest DPPH scavenging activity, with an IC50 value of 616 µg/mL, followed by tatsoi, with an IC50 value of 789 µg/mL. Further, the other four vegetables showed the lowest DPPH scavenging activity. Particularly, pink cabbage exhibited the lowest DPPH scavenging effect, with an IC50 value of 8,989 µg/mL. Moreover, the IC50 value of DPPH scavenging activity for the standard, ascorbic acid was 9.2 µg/mL.
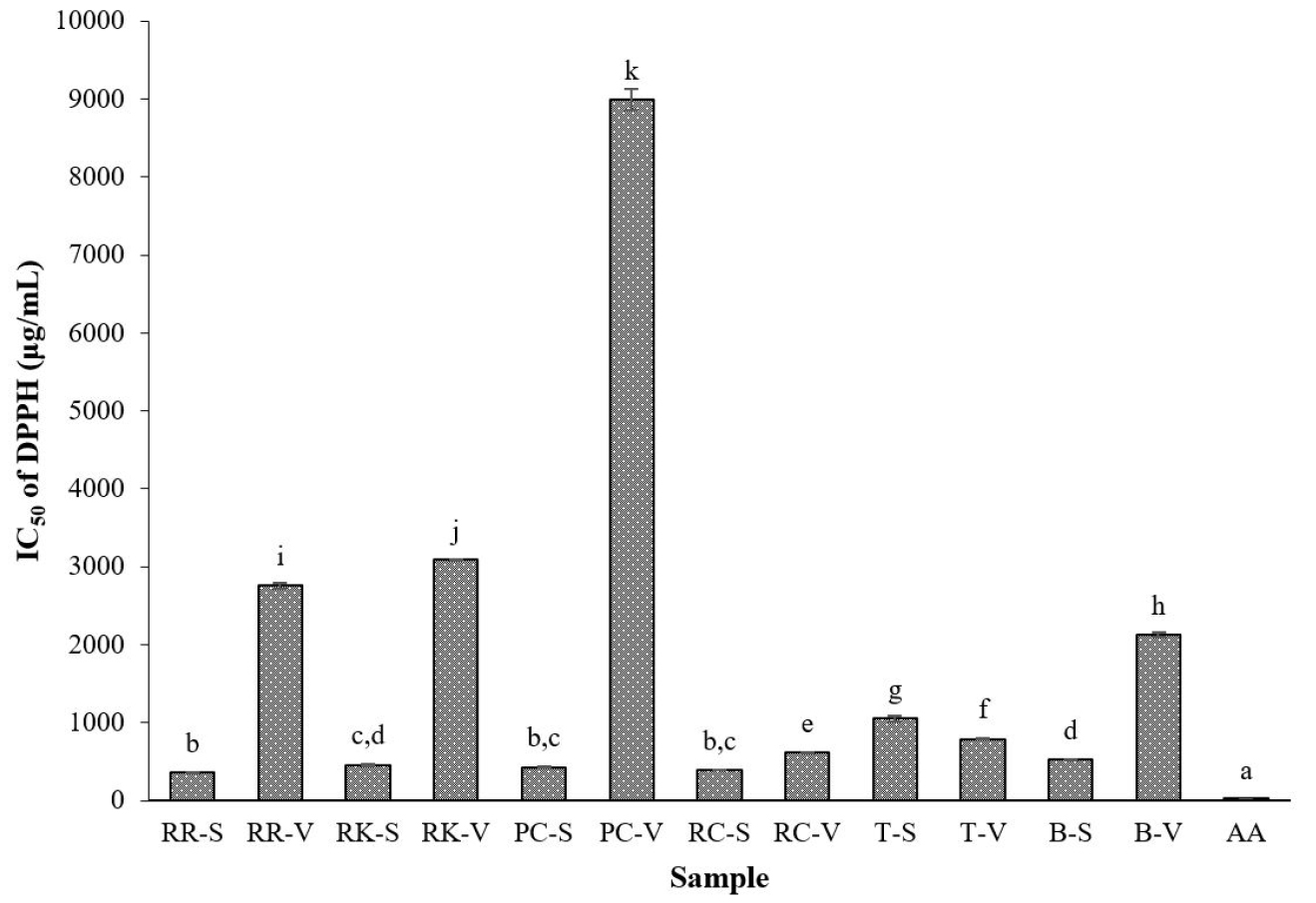
Fig. 1
1,1-Diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activity of Brassicaceae sprouts and their vegetables. Values are mean of three replicates (n = 3) ± standard deviation. Bars with different letters are significantly different (p < 0.05). RR-S, red radish sprout; RR-V, red radish vegetable; RK-S, red kohlrabi sprout; RK-V, red kohlrabi vegetable; PC-S, pink cabbage sprout; PC-V, pink cabbage vegetable; RC-S, red cabbage sprout; RC-V, red cabbage vegetable; T-S, tatsoi sprout; T-V, tatsoi vegetable; B-S, broccoli sprout; B-V, broccoli vegetable; AA, ascorbic acid.
ABTS•+ scavenging activity
Similar to the DPPH radical scavenging activity, the ethanol extracts of all of the sprouts showed higher ABTS scavenging activity than those of the vegetables, with the exception of tatsoi sprouts (Fig. 2). Regarding the sprouts, red radish registered the maximum ABTS scavenging activity, with an IC50 value of 413 µg/mL. Sprouts of pink cabbage, red cabbage, broccoli, red kohlrabi, and tatsoi also showed appreciable ABTS scavenging activities, with IC50 values of 450, 486, 584, 601, and 870 µg/mL, respectively. Regarding the vegetables, red cabbage and tatsoi exhibited the highest ABTS scavenging activity, with IC50 values of 578 and 587 µg/mL, respectively. Among the different vegetables, pink cabbage showed the lowest ABTS scavenging effect (IC50 of 8,304 µg/mL). The ascorbic acid standard registered a strong ABTS scavenging effect (IC50 of 18 µg/mL).
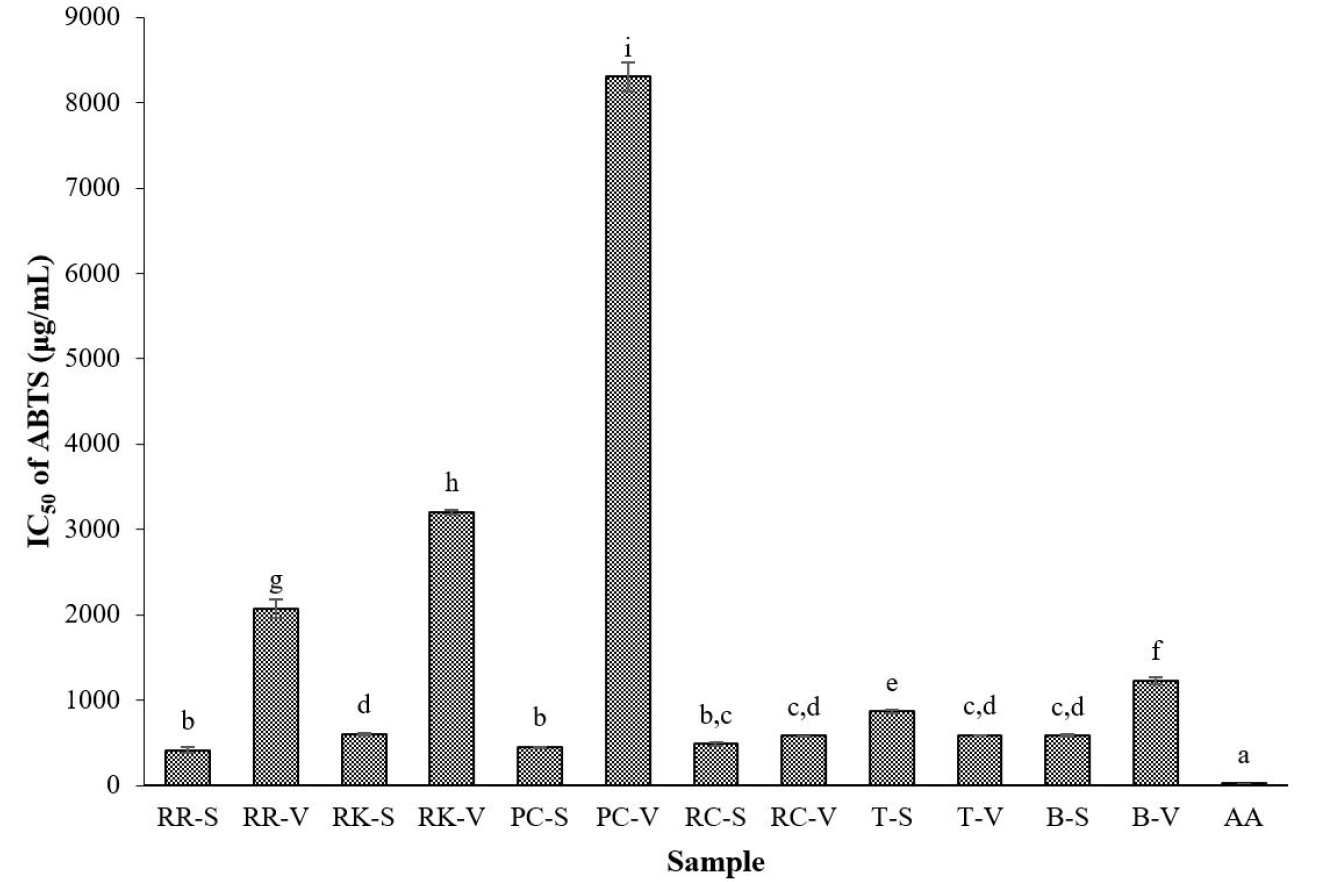
Fig. 2
2,2ʹ‐Azinobis (3‐ethylbenzothiazoline‐6‐sulfonic acid) diammonium salt (ABTS) radical scavenging activity of Brassicaceae sprouts and their vegetables. Values are mean of three replicates (n = 3) ± standard deviation. Bars with different letters are significantly different (p < 0.05). RR-S, red radish sprout; RR-V, red radish vegetable; RK-S, red kohlrabi sprout; RK-V, red kohlrabi vegetable; PC-S, pink cabbage sprout; PC-V, pink cabbage vegetable; RC-S, red cabbage sprout; RC-V, red cabbage vegetable; T-S, tatsoi sprout; T-V, tatsoi vegetable; B-S, broccoli sprout; B-V, broccoli vegetable; AA, ascorbic acid.
Discussion
Brassicaceae vegetables are one of the most important groups of low-carbohydrate foods. These vegetables are consumed by people worldwide, and are considered the main food crops in China, Korea, Japan, India, and many European countries. B. oleracea is the major vegetable species, and its cultivars include cabbage, broccoli, kohlrabi, cauliflower, and others (Cartea et al., 2011). Recently, the development of antioxidant-rich foods has gained considerable attention for the purpose of health promotion and disease prevention. The health-promoting effects of Brassicaceae vegetables are mainly ascribed to their complex mixture of bioactive metabolites, which have strong antioxidant potential (Vale et al., 2014). The antioxidant potential of Brassicaceae vegetables is associated with their phenolic content. Brassicaceae sprouts are richer in phenolic compounds than their respective commercial vegetables (Pajak et al., 2014). Hence, a comparison of the phytochemical composition and antioxidant activities of Brassicaceae sprouts with those of their commercial vegetables is key to developing new functional foods (Nicola et al., 2013).
The results of DPPH and ABTS scavenging activities in the present investigation of Brassicaceae sprouts and their commercial vegetables were compared and correlated with each other. The total phenolic content exhibited a strong correlation with total flavonoid (R2 = 0.9487) and moderate correlation with DPPH (R2 = 0.4983) and ABTS (R2 = 0.5032). Further, there was a close correlation between ABTS and DPPH (R2 = 0.9831). However, there was no significant correlation between tannin content and antioxidant activity.
In this study, we determined the total phenolic, flavonoid, and tannin contents of six Brassicaceae sprouts (red radish, red kohlrabi, pink and red cabbage, tatsoi, and broccoli) and their respective vegetables. Additionally, we investigated the antioxidant activity of sprouts and vegetables using DPPH and ABTS assays. The sprouts of all six plants contained higher total phenolic and flavonoid contents than their commercial vegetables, except for tatsoi sprouts. Apart from broccoli sprouts, the other sprouts contained very low tannin contents. Polyphenolic compounds are involved in a variety of biological functions. It is well-known that phenolic components, including phenolic acids, flavonoids, anthocyanins, and proanthocyanidins, may regulate numerous signaling pathways involved in the survival, growth, and differentiation of cells (Cartea et al., 2011).
DPPH and ABTS radical scavenging activities have been extensively used to examine the antioxidant activity of plant products, mainly to assess the ability of chemicals as free radical scavengers or hydrogen donors (Sowndhararajan and Kang, 2013). In this study, all sprouts scavenged the DPPH and ABTS radicals more effectively than did their respective vegetables, with the exception of the tatsoi vegetable, which provided a better scavenging effect than its sprouts. Among the different sprouts, those of red radish showed the highest DPPH and ABTS scavenging activities. The IC50 values of DPPH and ABTS assays were 358–1,053 and 413–870 µg/mL for sprouts, and 616–8,989 and 578–8,304 µg/mL for vegetables, respectively. Among different vegetables, pink cabbage registered the lowest total phenolic and flavonoid contents and antioxidant activities. However, we found higher levels of total phenolic and flavonoids in pink cabbage sprouts, as well as considerable antioxidant activity. As well as their high total phenolic and flavonoid (known for their antioxidant capacity) contents, the sprouts showed higher DPPH and ABTS radical scavenging activities.
There are several studies that evaluate the phytochemical composition and biological activities of Brassicaceae sprouts (Baenas et al., 2012; De Nicola et al., 2013; Samec et al., 2018; Vale et al., 2014; 2015; Wei et al., 2011). In a recent study, Samec et al. (2018) investigated the phytochemical composition and the activity of endogenous enzymes in five Brassicaceae sprouts: white cabbage, kale, broccoli, Chinese cabbage, and arugula. In their study, white cabbage sprouts showed the highest amount of polyphenols (18.34 mg GAE/g dry weight [DW]) and glucosinolates, as well as a strong antioxidant activity, followed by kale sprouts. In another study, Vale et al. (2014) studied the total phenolic content and antioxidant activity of B. oleracea sprouts such as broccoli, Portuguese Galega kale, Portuguese Tronchuda cabbage, and red cabbage using DPPH, hydroxyl, and peroxyl radical scavenging and ferrous ion chelating ability assays. These four B. oleracea sprouts exhibited significantly different antioxidant activity; red cabbage grown under light cycles exhibited the highest antioxidant activity. The authors found that the antioxidant capacity of sprouts declined with sprouting and increased in the presence of light. Baenas et al. (2012) performed the phytochemical profiling of Brassicaceae varieties such as broccoli, kohlrabi, red cabbage, rutabaga, turnip, turnip greens, radish, garden cress, and white mustard. They found that the levels of glucosinolates in seeds were significantly higher than those in sprouts, and considered eight-day-old sprouts as optimum for consumption. Sprouts from red cabbage and radish presented the highest total glucosinolate and total phenolic contents, biomasses, and antioxidant capacities.
Glucose treatment significantly improved the total phenolic, total glucosinolate, and anthocyanin contents, as well as ferric ion reducing antioxidant power, in Chinese kale and pak choi sprouts. In radish sprouts, glucose treatment enhanced only the anthocyanin and ascorbic acid contents (Wei et al., 2011). De Nicola et al. (2013) determined the phytochemical compositions of two B. oleracea varieties (broccoli and Tuscan black kale) and two R. sativus varieties (Daikon and Sango). Among these, Sango sprouts showed the highest total phenolic (19.4 mg caffeic acid equivalent/g DW of sprouts), flavonoid (9.7 mg catechin equivalent/g DW of sprouts), and anthocyanin (5.8 cyanidin 3-glucoside equivalent/g DW of sprouts) contents. Vale et al. (2015) reported that the levels of bioactive compounds in the sprouts from red cabbage, broccoli, Galega kale and Penca cabbage significantly changed after seven days of storage. The results from previous studies clearly demonstrated that the germination time (days) and extraction solvent highly influenced the polyphenolic content and antioxidant activity of the sprouts. Furthermore, the actual chemical composition of the seeds essentially changes during germination.
Heimler et al. (2006) investigated the radical scavenging activity and total phenolic, flavonoid, and total condensed tannin contents of seven Brassicaceae vegetables (Italian kale, broccoli, Savoy cabbage, white cabbage, cauliflower, green cauliflower, and Brussels sprouts). The results showed that the EC50 values ranged from 81.45 (broccoli) to 917.81 (cauliflower) mg sample/mg DPPH, and the total phenolic content ranged from 4.30 (Savoy cabbage) to 13.80 (Italian kale) mg GAE/g DW. Further, Jaiswal et al. (2011) investigated the total phenolic content, antibacterial and antioxidant potentials of 60% ethanol, acetone, and methanol extracts of B. oleracea vegetables (broccoli, Brussel sprouts, and white cabbage). The results revealed that the methanol (60%) extract registered the highest polyphenolic content for broccoli (23.6 mg GAE/g extract), Brussels sprouts (20.4 mg GAE/g extract), and white cabbage (18.7 mg GAE/g extract). In this study, the total phenolic content in the ethanol (95%) extracts of broccoli sprouts and vegetables were 25.750 and 20.293 mg GAE/g extract, respectively. The solvent used for extraction also plays a major role in the total phenolic content and antioxidant activity. Sprouts, which can be consumed in their fresh form in all seasons, are an excellent substitute for plant foods. Further, sprouts are inexpensive, easy to grow, and fast growing. The results of this study also confirmed that sprouts from edible plants are a good source of antioxidants.
Conclusion
This study found highly varied total phenolic and flavonoid contents and antioxidant activity of Brassicaceae sprouts and vegetables. The results of our investigation indicated that five Brassicaceae sprouts (red radish, red kohlrabi, pink cabbage, red cabbage, and broccoli) contained higher levels of polyphenolic components with appreciable antioxidant properties than did commercial vegetables. We concluded that sprouts are a rich source of natural antioxidants and can be used as plant-based foods.




